文章: The Professional Buyer’s Guide to Red Light Devices

The Professional Buyer’s Guide to Red Light Devices
In the Red Light Therapy market, "Third-Party Independent Testing" is a common marketing phrase. But it leaves one critical question unanswered: If a brand has already passed the tests, why haven't they applied for Medical Device Clearance?
The "Regulatory Gap": A Hidden Industry Standard
Third-party testing is a one-time "snapshot." It simply means a lab looked at one device on one day. It does not require the company to maintain quality standards, report adverse effects, or prove clinical efficacy over time.
Applying for a Class II Medical Device designation (FDA, CE, TGA, Health Canada) is the ultimate commitment to consumer safety. It requires:
- Legal Accountability: Mandatory audits of manufacturing facilities.
- Post-Market Surveillance: Legal obligation to report any malfunctions or injuries.
- Clinical Substantiation: Rigorous proof that the device delivers the therapeutic results promised.
The Crucial Question for Any Brand:
"If your technology is safe and effective enough to pass a lab test, why haven't you submitted those results to the FDA for official clearance?"
The answer is often that regulatory bodies demand a level of manufacturing precision and clinical evidence that most "gadget" companies cannot meet.
5 Scientific Factors That Differentiate Celluma
Celluma doesn't hide behind vague lab reports. We are a globally regulated medical tool because we adhere to the highest standards of Photobiomodulation (PBM) science:
1. Patented "Shape-Taking" Design
Physics dictates that light energy diminishes rapidly with distance (The Inverse Square Law). Celluma’s flexible design allows for direct skin contact, ensuring maximum photon absorption by the mitochondria.
2. Pulsed Delivery (PWM)
Continuous light can cause cellular "saturation." Celluma uses Pulse Width Modulation to give cells the micro-rests needed to maximize ATP production and prevent overheating.
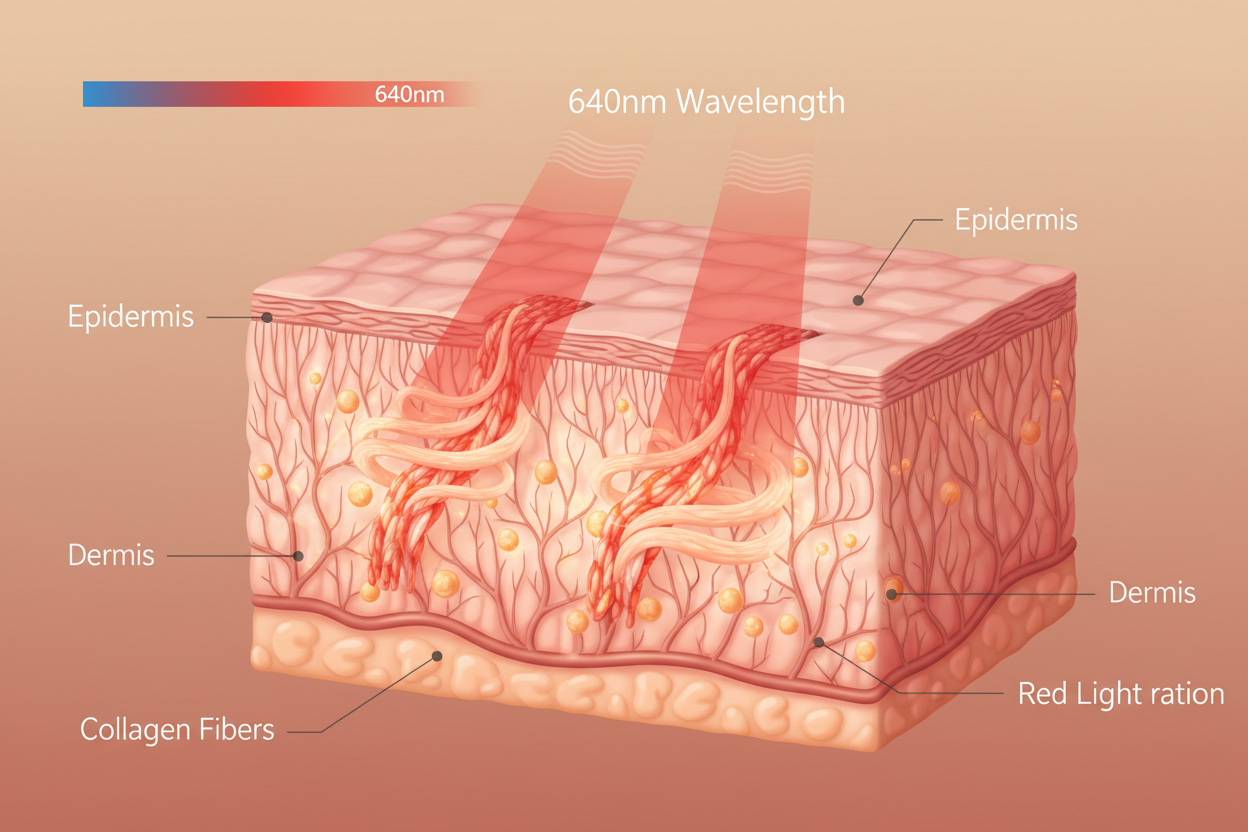
3. Clinical Wavelength Precision
We use specific, high-intent wavelengths: Blue (465nm), Red (640nm), and Near-Infrared (880nm). Each is verified by medical regulators for its specific biological target.
4. Uniform Energy Distribution
Unlike panels that have "dead zones" at the edges, Celluma is engineered to provide a perfectly uniform dose across the entire treatment area.
5. Verified Biocompatibility
As a medical device, every material used—from the LEDs to the outer coating—is tested for safety and non-toxicity, a standard consumer-grade gadgets rarely meet.
Don't Settle for "Testing" Without Accountability
Invest in the medical-grade standard trusted by clinics worldwide.
Invest in Celluma Medical-Grade LEDScientific Verification Center
📖 Why Celluma? – The 4 key reasons to choose our technology.
📖 Mechanism of Action – What happens during a 30-minute session.
📖 Mitochondria & ATP – How your cells use light for energy.

发表评论
此站点受 hCaptcha 保护,并且 hCaptcha 隐私政策和服务条款适用。